Informacje o artykule
DOI: https://www.doi.org/10.15219/em99.1612
W wersji drukowanej czasopisma artykuł znajduje się na s. 89-98.
 Abstract in English
Abstract in English
Jak cytować
Marciniak, M. (2023). Key productivity factors in drug discovery and development projects. e-mentor, 2(99), 89-98. https://doi.org/10.15219/em99.1612
E-mentor nr 2 (99) / 2023
Spis treści artykułu
- Abstract
- Introduction
- Productivity of pharmaceutical R&D
- R&D drug discovery budgets and spending
- Overview of the drug discovery process
- Pharmaceutical R&D risk management
- Methodology
- Results
- Conclusions
- Acknowledgements
- References
Informacje o autorze
Key productivity factors in drug discovery and development projects
Magdalena Marciniak
Abstract
The field of health care is an important element of the economic and social life of every country in the world combining demographic, economic and epidemiological, ethical and social challenges. Spending on the development of new therapies has been increasing over the past two decades, and the amount of drugs approved by regulatory agencies has remained stable. Literature does not provide adequate knowledge about the reasons of the productivity drop that impacts the competitive advantage of the companies taking part in the project’s race to the market (Schuhmacher et al., 2022), and it therefore seems crucial to analyse the factors determining high productivity of the pharmaceutical industry to adjust further actions ensuring the highest quality of health care systems, focusing on the wellbeing of the patient and the development of increasingly safer medicines. To address this need the author performed systematic literature review followed by structured interviews with 14 experts working globally in the field of drug development to determine productivity factors in drug discovery research and development projects, with the goal of answering questions related to which factors play a key role in the productivity of scientific organisations and the relationship between the factors, providing an insight into which parts of drug discovery ecosystem can increase a chance to address highly unmet medical needs of patients waiting for novel, safe and effective forms of treatment. As a result of the research 22 key productivity factors were defined and clustered into 4 categories: scientific, managerial, business, environmental and relations between the factors were discussed.
Keywords: drug discovery, drug development, innovation, productivity, R&D, project management
Introduction
The field of healthcare is an important element of the economic and social life of every country in the world. Pharmaceuticals account for a significant portion of total healthcare expenditure in developed countries, and for this reason Western European countries, the United States, Canada, Japan, Australia, New Zealand, Israel, Singapore and South Korea annually spend a significant portion of their GDP (gross domestic product) on investments in research and development projects - a figure ranging from 2.5% to 4.5% (Mikulic, 2021). Entities operating on the healthcare market are subject to numerous and variable regulations aimed at responding to the needs of the physical and mental safety of patients, respect for the dignity of people and animals, as well as the potential long-term effects of the use of therapy for future generations. In addition, the functioning of these entities takes place on the principles of free competition aimed at maximising profits, developing innovation and minimising resource losses. Over the past two decades, spending on the development of new therapies has been increasing, while the amount of drugs approved by regulatory agencies has remained stable. An observed decrease of productivity in the pharmaceutical industry led to the need to analyse factors influencing the process of medicine development and defining key success factors in drug discovery and development projects (Bode-Greuel et al., 2008, Bukowski, & Gierczyński, 2019). High competition on the market makes companies reluctant to share know-how and methods of drug development, so the literature does not provide adequate knowledge about the productivity of drug development projects. It therefore seems crucial to analyse the factors determining the productivity of the pharmaceutical industry to adjust further actions ensuring the highest quality of functioning of the components of the healthcare system, focusing on the wellbeing of patients and the development of increasingly safer medicines. To address this need the author performed a deep literature review followed by structured interviews with 14 experts in the field of drug development in order to determine productivity factors in research and development projects in the field of biotechnology and pharmacy.
Productivity of pharmaceutical R&D
R&D productivity can be defined as the relationship between the value created by a drug - commercial and medical - and the investments required to generate the medicine. In the literature R&D productivity is defined as the ability of R&D to translate input, such as investments, into output, such as the number of approved drugs or other defined milestones (Paul et al., 2010).
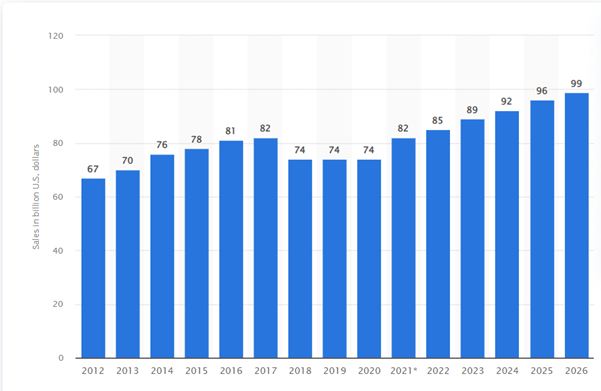
In the literature there is emphasis on the decline in revenues owing to the expiration of patents, a phenomenon correlated with the lowering number of truly innovative molecules (both small molecules and biologics) recently approved by the Food and Drug Administration (FDA) (FDA, 2023). On the one hand more demanding requirements of regulators driven by safety and efficacy can be observed, while on the other - issues related to the transparency of the data and reproducibility, which cause higher requirements of the agencies. These two aspects are strongly correlated to each other and reflect a decrease in the quality of robust data in the drug discovery and development industry (Schuhmacher, 2022, Stalder, 2022). A lower number of novel drugs with a long patent life is reflected in the statistics and predictions shown by Statista.com, visualising the total global generic prescription drug revenue from 2010 to 2024 (in billions of US dollars) and predicting an increase in the sales of generic drugs (Figure 1). It suggest that a limited number of new medicines will be available and the current market standard will play a key role in the treatment of patients (Mikulic, 2022; Mullard, 2023).
Figure 1
Total worldwide generic prescription drug revenue 2012-2026
Source: Worldwide generic prescription drug sales 2012-2026, M. Mikulic, 2022 (https://www.statista.com/statistics/309411/global-total-generic-prescription-drug-revenue/).
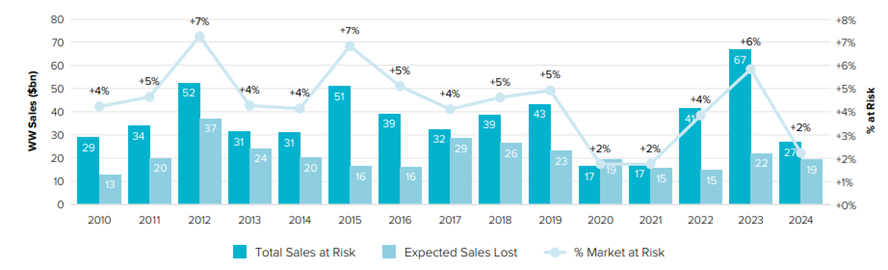
Additionally, in the Evaluate report from 2018 the authors present sales at risk caused by the expiration of patents for the years 2010-2024, shown in Figure 2.
Figure 2
Worldwide sales at risk from patent expiration (2010-2024)
Source: World Preview 2018, Outlook to 2024 (11th ed.), 2018. EvaluatePharma. (https://www.evaluate.com/sites/default/files/media/download-files/WP2018.pdf).
These statistics depict the total generic prescription drug revenue worldwide from 2010 to 2024. By 2024 the global prescribed generics market is expected to grow to 100 billion US dollars. The loss of profits from generic drugs, and the resulting relatively lower investments in R&D, highlight the need for the industry to improve productivity and maintain risk management of R&D divisions to replace old treatments with new, more effective medicines approved by the FDA and other regulatory agencies operating worldwide (Stalder, 2022).
Productivity and investments are correlated with the number of projects performed by drug discovery companies and project management models used to deliver drugs to patients (Schuhmacher et al., 2021).
A project is defined as a temporary effort to create a new product or service that cannot be achieved through standard processes and operations, with a definite beginning and end. It is an unusual activity, burdened with uncertainty, and therefore bears increased risk compared to typical, everyday operations (Mingus, 2002).
Project management is the use of specific knowledge, methods, skills, tools and techniques aimed at delivering an outcome to a project client (Project Management Institute, 2021, p. 12 ).
Research and development projects aimed at the development of therapeutics have a high degree of complexity and variability, long duration, difficult-to-determine product profile and unknown target audience. In such projects, there is work underway in parallel on the product - the drug, and on the research methods needed to create it (Schuhmacher, 2022).
R&D drug discovery budgets and spending
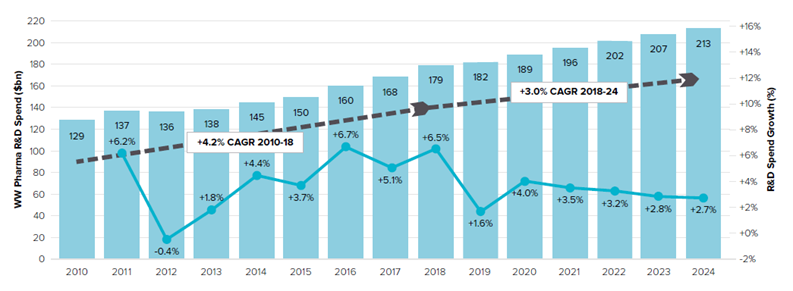
A report published by The EvaluatePharma in 2018 presents data suggesting a worldwide increase in R&D spending in the pharmaceutical industry (Figure 3). Global R&D spending in 2017 increased by 3.9% to 165 billion US dollars compared to 2016. As calculated, in 2017 the top 20 pharma companies spent a total of 97.2 billion US dollars, with Roche as the top spender with 9.2 billion US dollars investment in R&D in 2017. As in previous years, R&D intensity remains highest in the United States, with more than half of the global R&D expenditure utilised by US pharma giants. The authors of the report also state that overall R&D spending is expected to grow by 3% year-by-year, and predict it will reach around 204 billion US dollars by 2024 (EvaluatePharma, 2019).
Figure 3
Worldwide total pharmaceutical R&D spending in 2010-2024
Source: World Preview 2019, Outlook to 2024 (12th ed.), 2019. EvaluatePharma. (https://info.evaluate.com/rs/607-YGS-364/images/EvaluatePharma_World_Preview_2019.pdf).
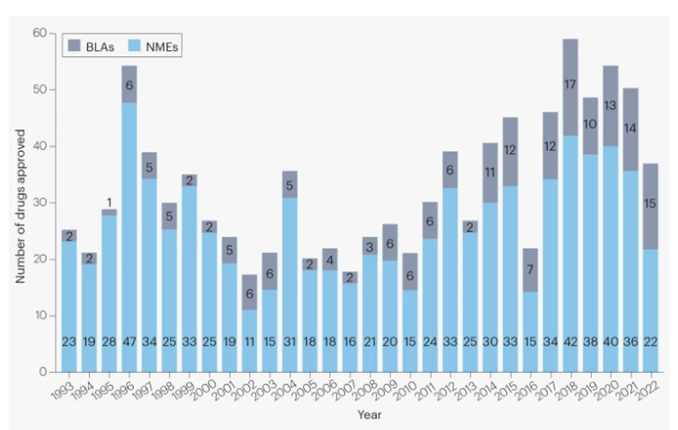
The results of this investment are visible in the number of new FDA approvals - in 2016 the FDA approved 27 new molecular entities, followed in 2017 by 46 drugs, then 59 in 2018, 48 in 2019, 53 in 2020, 55 in 2021, and 37 in 2022 (FDA, 2023; Mullard, 2023).
Figure 4
Novel FDA approvals since 1993
Source: "2022 FDA approvals", A. Mullard, 2023, January 3. Nature Reviews Drug Discovery, 22, p. 83 (https://doi.org/10.1038/d41573-023-00001-3).
The high number of drugs with ongoing late-stage development and approval phases contribute to the increased average cost of drug development over the last years. Therefore, although the number of new drugs is relatively stable (Figure 4), the cost of discovery and development is much higher, and paradoxically - the productivity of the market is decreasing (Schuhmacher et al., 2022). The authors of the Evaluate report also conclude that despite an initial peak of investment in 2019, the proportion of R&D spending compared to pharmaceutical revenue decreased significantly in subsequent years. An increased number of drugs in late-stage development and the number of approvals contribute to an increased average cost of drug development, as Phase 3 trials remain the costliest step (Ng, 2015). Nowadays, in the discovery and development process researchers are able to use various tools aimed at more detailed and reliable data, and it is common practice to confirm data in different and more sophisticated assays or models to avoid misinterpretation and increase the probability of success. This also leads to higher investments in the earlier stages of development and, as a consequence, increase spending on R&D. Despite more available tools for early stage discovery and the higher number of molecules reaching clinical trials, the number of approved drugs remains stable (Kiriiri et al., 2020; Yeung et al., 2021).
In a 2014 paper, together with colleagues Cook presents findings on the main scientific and technical determinants of project success and the high quality of the Astra Zeneca pipeline defined as the five Rs, listed below (Cook et al., 2014):
- the right target - defined as a solid understanding of biology and confirmation of the link between the selected target and a disease of interest,
- the right tissue - the candidate molecules developed must exhibit exposure and pharmacological activity in the selected tissue or organ,
- the right safety - the right safety profile in the selected indication is one of the key success factors for the drugs. Additionally, any safety issues should be analysed at each, even very early, stage of development,
- the right patient - selection of the right patient profile is based on an understanding of the current standard of care and how certain aspects of the patient's condition may affect their response to the drug, bearing in mind drug-drug interactions, gender or other diseases,
- the right commercial potential - this is understood as fast delivery of medical differentiation after the drug reaches the market, in consequence securing commercial value.
The authors emphasise the need for scientific curiosity confirming the research mindset and avoiding a "volume-based" approach, therefore promoting transparency by asking and answering "killer questions" when deciding whether to move assets to the next stage.
The literature points to several potential issues that may cause a decrease in the productivity of R&D drug discovery work (Antonijevic, 2015; Cook, 2014; Czech, 2022; Schuhmacher et al., 2022):
- easy-to-make drugs have already been discovered, and the current structures of the drugs are more challenging,
- the broad patenting culture of companies, blocking other players from developing drugs,
- the industry is working on more complex and sophisticated disease mechanisms, which require more detailed work,
- frequent changes of regulations and the reserved approach of regulators to the risk related to new drugs approval. This leads to increased costs of clinical trials due to the regulations of agencies and the increased number of patients enrolled during each phase,
- a challenging reimbursement and payer environment also escalates the cost of clinical trials.
The above all show that the mindset of R&D research has shifted and become more commercialised or industrialised. Bearing in mind that only 1 out of 10 projects that reach clinical trials will meet the market, the industry is under pressure to increase the number of projects under development. As a consequence an increase in the number of projects can be observed, but with a stable number of approved treatments. Due to market needs, business models and financial costs, scientists are more focused on corporate goals to deal with the amount of projects that end up in the clinics, and are not or cannot be strongly focused on the quality of the assets, including knowledge of the issue at hand, its relation to specific diseases and its therapeutic potential.
To better understand the complexity and risk associated with development of new drugs it is good to take a general look at the process of drug discovery and development.
Overview of the drug discovery process
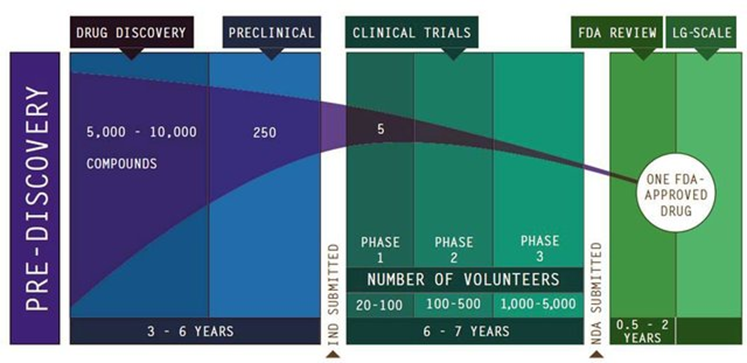
Innovative and new drugs that fulfill unmet medical needs are the key value drivers of research-oriented companies from the pharmaceutical industry (Schuhmacher, 2016). Each drug research and development (R&D) programme can be considered as a long-lasting and highly complex process that requires cooperation between multiple disciplines. The typical work-flow of the drug research and development process is illustrated in Figure 5 and consists of the following stages (Chen et al., 2018):
Figure 5
Stages of the drug research and development process
Note. Attrition rates of compounds at subsequent stages of the drug development process are presented - from approximately 10,000 candidates to only one compound making it to the market.
Source: "Pharmaceutical R&D", D. W. Light, 2012. The Politics of Medicines (e-Encyclopaedia), p. 8 (https://haiweb.org/encyclopaedia/pharmaceutical-
research-and-development/).
The general drug development process can be divided into several steps (Chung et al., 2015; FDA, 2018; Ng, 2015; Poduri, 2021, Rydzewski, 2008):
Step 1: Target Identification & target validation - The first step in the drug development process involves discovery work, and is where drug development companies choose a molecule, such as a gene or protein, to target with a drug.
Step 2: Hit identification - This is the stage where compounds that may be able to reach the target are identified.
Step 3: Hit to lead (H2L, hit confirmation) - This step is related to confirmation of the compounds found, using several methods and ways to confirm that the company can work on the compounds.
Step 4: Lead optimisation - At this stage compounds are tested in animal models to translate and confirm results generated in vitro. If necessary, the chemical structures of the lead compounds can be altered to improve their selectivity and specificity towards a given target.
Step 5: Preclinical testing - Here work is performed on the compound(s) selected as potential clinical candidates. Aspects related to chemical parameters of the active compound and formulations are tested. Advanced in vitro and in vivo testing has to be performed.
Step 6: Investigational New Drug (IND) application filing - The third step involves submitting an Investigational New Drug application to the FDA prior to beginning human clinical trials.
Step 7: Phase 1 clinical studies - The first phase of human clinical testing involves a relatively small group of patients (healthy volunteers or with developed diseases) and focuses entirely on safety.
Step 8: Phase 2 clinical studies - The patient pool widens to 100 or more patients, and the patients are afflicted by the disease. Safety remains a big focus of phase 2 studies, with short-term side effects closely monitored, although increasing emphasis is starting to be placed on whether or not a drug is working as expected and if it is improving the condition of the subjects under study.
Step 9: Phase 3 clinical studies - Safety remains a priority here, but efficacy also plays a big role. Phase 3 studies are designed by drug developers but approved by the FDA, with guidelines for a clearly defined primary endpoint to determine the success or failure of a tested drug. Phase 3 trials involve even more patients, perhaps a few hundred to maybe thousands, and are by far the longest and costliest components of the drug development process.
Step 10: New Drug Application filing - This step focus on filing a New Drug Application with a chosen regulatory agency, and contains all research and safety data examined during the prior steps. If approved, the drug becomes immediately available for commercial production.
Step 12: Phase 4 clinical studies - Following approval, agencies can request long-term safety studies to be undertaken whereby drug developers are required to submit regular reports detailing any adverse events with the drug to the agency.
Pharmaceutical R&D risk management
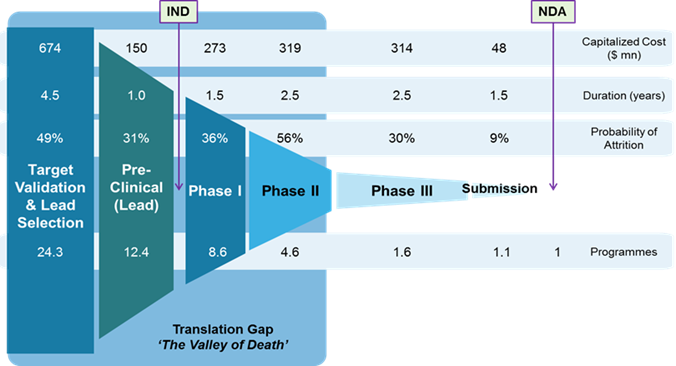
The drug discovery and development programme is a long-term, high-risk, high-cost but also high-benefit type of business activity (Chen et al., 2018). On average, it takes over 12 years for the programme to get from target identification to marketing approval (DiMasi, 2000) at an overall cost of about 2.6 billion US dollars. As summarised in more detail in Figure 6, the lengthy duration and high costs of programmes are accompanied with the high probabilities of drug candidate attrition throughout the entire process. Despite the high-risk and high-cost attributes, from the business perspective drug R&D programmes should still be considered as high-reward initiatives.
Figure 6
The drug development lifecycle with (i) duration of stages, (ii) capitalised costs (in 2010, in US dollars) and (iii) probability of failure
Source: "Developability assessment as an early de-risking tool for biopharmaceutical development", J. Zurdo, 2013, (Pharmaceutical Bioprocessing), 1(1), p. 30.
The overall failure rate in drug development is over 96%, while at the clinical trial stage, when the drug is administered to humans, the figure is 90%. The rates are highest for drugs developed for new disease entities or for diseases that are currently incurable (Hingorani et al., 2019). The average cost of introducing a new drug to the market is 2 billion US dollars (Sagar, 2017). New medicinal substances were traditionally obtained from natural products, but with the development of chemical knowledge, technology and the growing demand for medicinal substances, industry interest has been redirected to high-performance synthesis and development based on chemistry, biochemistry and combinations of various sources of potential medicinal substances (Berdigaliyev et al., 2020; Schuhmacher et al., 2021). High competition in conducting research and development work forces companies to patent early, even when the first results appear, indicating the therapeutic potential of the compounds under development, i.e. at the stage of discovery work. This reduces the freedom of action of scientists in terms of the possibility of developing already patented compounds (so-called freedom to operate), hence the competition and pressure for rapid development and patent protection of potential drugs is still growing (Antonijevic, 2015; Stalder, 2022).
In order to increase the chances of introducing a drug to the market, companies simultaneously implement many research and development projects and invest in technologies supporting their development, which additionally increases the average cost of drug development. However, as previously discussed, the increase in the research budget, the development of new technologies, and a better understanding of molecular biology do not correlate with the number of newly registered drugs (Liberti et al., 2010; Petro & Gardiner, 2015; Wallmark, 2016).
Methodology
In order to define the main risk areas and productivity factors in projects focused on drug discovery and development, an in-depth systematic literature review was performed. For the literature review in the Scopus database the following key words were used: drug discovery, drug development, productivity, project management, taking into account the areas of pharmacy, biotechnology, medicine, business and economics, and 3187 articles were obtained during the research. Selected articles were reviewed in order to define key areas of research project management and practices in biotechnology and drug development. This approach is in line with the general principles of systematic literature review (Tranfield et al., 2003). Industry guidelines in biotechnology, drug development and registration were also analysed, taking into account: guidelines of leading regulatory agencies; good practices: GLP - Good Laboratory Practices, GMP - Good Manufacturing Practices, GCP - Good Clinical Practices; legal acts related to the development of medicines, including but not limited to patent law, drug registration processes and guidelines of bioethics committees.
Based on the outcome of the literature review, the author prepared a list of 37 questions and conducted structured interviews with 14 experts in the scientific and managerial areas of drug discovery and development, e.g. key areas of drug development, necessary information needed for decision-making, tools used to identify risks, and the decision-making process in the company, with interviews conducted online and in person from May 2022 till August 2022. Interviews as a research method were chosen in order to learn about the project management system in more detail, elucidate data available in the literature and draw on many years of experience of the responders from various R&D projects. The interviews were than transcribed and coded. The author defined 22 factors influencing the productivity of drug discovery projects and grouped them into four categories: scientific, managerial, business and environmental.
Results
During the literature review and interviews, 22 key factors influencing risk and the success rate of projects in the area of new drug development were recognised and analysed. Factors were grouped as scientific, managerial, business and environmental, and presented in Table 1 according to the frequency and importance given in the literature review and interviews.
Table 1
Groups of key productivity factors in drug discovery and development projects
| 1. Scientific factors | 2. Managerial factors |
|
|
| 3. Business factors | 4. Environmental factors |
|
|
Source: autor's own work.
Scientific factors:
Complexity of diseases and pathophysiology - despite the rapidly developing science, we still no little about most disease and pathophysiological processes taking place in the human body. Even in well-known areas, such as infectious diseases, where the knowledge of disease development is quite broad and detailed, new diseases and mechanisms of disease development appear, posing challenges for modern pharmacology. Understanding the disease process is an important success factor in rational drug discovery and development. New disease entities are often multifactorial, which entails the body's compensatory defense mechanisms and, consequently, the appearance of side effects.
Molecular target selection - the molecular target is selected on the basis of available data confirming its role in the development of the disease. Basic research data is often incomplete, meaning the decision to choose a target and start a project is based on uncertainty. At the same time, the competitive environment is analysed, which often defines whether work on even an interesting and adequate molecular target will be undertaken.
Selection of a compound for further development - the development of drugs is very expensive, hence companies can afford to develop few or only one compound that has a chance of becoming a drug. Thus, the choice of the molecule to be developed simultaneously determines which compounds will not have a chance to be used by patients.
Selection of patient populations - patient populations are often conditioned by the properties of the compound that has been developed, a decision that is made a few years after starting work on the compound, and one that is defined by its pharmacodynamic properties. The larger the population of patients who respond to therapy, the greater the likelihood of a return on investment. The development of drugs for rare diseases is therefore burdened with a high investment risk, and often abandoned.
Novel methods and design strategies - the development of technologies supporting the characteristics of the drugs under development, reducing the cost of the initial stages of drug discovery and development, and accelerating the entire process, has resulted in many companies choosing to simultaneously deal with multiple innovative molecular targets, increasing the pool of potential compounds from which the most promising for further development are then selected.
Biomarkers availability - responders emphasise the need to start work on biomarkers early, already at the stage of the lead compound or, if possible, even earlier in order to increase the probability of success of the projects. This approach affects both the comfort of study participants and the staff involved in conducting research, but also allows for minimising the number of errors that may affect clinical results.
Managerial factors:
Involvement of the main stakeholders when making decisions regarding initiation of the project - the study indicates a multidimensional analysis of the project assumptions as an important aspect affecting the success of the project. When making a decision to start work on a selected drug, it is important to involve people with knowledge of various areas of drug development - including those who will actively participate in the project in a few years, e.g. the regulatory department, clinical department, business development department, marketing.
Appropriate preparation for project management - experts emphasised the need for substantive preparation and experience of people related to project management, including project managers and people from senior management, with many of them particularly emphasising the impact of the project manager's competence on the effectiveness of communication, enforcement of tasks in the project, structuring of work on the project, and the preparation of appropriate and transparent project documentation that will be an appropriate source of information for years to come. Respondents emphasised the many years of work on the project to come, and the need to refer to results generated several years earlier, which are no longer legible and understandable after the this amount of time.
Career paths for scientists - drug development is driven by scientists working in laboratories, who set the pace and direction of work in the course of their daily duties thanks to their commitment, knowledge and motivation. A lot of the knowledge and understanding of the relationships between the different areas of the project being developed is in their minds, hence responders recognised the need to shape appropriate scientific career paths supported by an appropriate level of wages and prestige satisfaction in order to maintain the pace and proper direction of drug development. In many organisations it is more highly rewarded and prospective to take on an administrative or managerial position than a scientific one. In addition, managerial development paths are often the only possible opportunities for promotion for researchers, which changes the possibility of substantive involvement in the project. An important aspect raised by the respondents was the fact that scientists were promoted too quickly, especially in small biotechnology companies. Inexperienced scientists are given higher, decision-making positions, as it is difficult for biotechnology companies to compete for resources with large pharmaceutical companies. People with little experience then face the need to make important decisions related to the project, while lacking the experience to do so.
Scientific cooperation - the pharmaceutical market is inherently very competitive, since any company that manages to be the first to introduce an innovative drug to the market receives exclusive sales. Cooperation on new drugs seems reasonable, allowing to reduce costs incurred for high-risk projects, increase the resources and pool of competences involved in the development of new medicines, and diversify risks and invest in other projects. The pharmaceutical industry uses many models of cooperation based on the relationships of companies, research centres and academia, as well as public-private partnerships in various configurations.
Outsourcing - companies can outsource the whole process, selected stages or specific tasks that aim to accelerate, reduce costs or access new competences and technologies. The field of outsourcing in the pharmaceutical industry has grown significantly in recent years in China and India, where several contract research organisations (CROs) are based, supported by cheaper labour, cheaper land and extensive laboratory infrastructure. There is also a phenomenon of strategic outsourcing based on the long-term, often exclusive, cooperation of drug development companies with selected service providers.
Appropriate, complete and clear project documentation - during drug development, not only is a therapeutic compound created, but also a wealth of information necessary or useful for clinicians and regulatory agencies to decide whether to start clinical trials or authorise them for use. An important aspect of the work on the drug, therefore, is to conduct accurate, transparent and complete design documentation, which is, in a way, a second outcome of research work.
Selection of a comparative compound (benchmarking) - early comparison of our potential drug to other leading therapeutics used as a standard form of treatment, as well as to competing compounds at the discovery stage developed by other research groups, allows us to verify the benefits of using our therapy.
Business factors:
Ensuring a long-term and flexible financing strategy - appropriate resources and a research budget adequate to the stage of development and the competitive environment allow for appropriate planning and decisions related, among others, to the number of diseases for which the potential drug is being developed, the clinical trial plan, but also about the forms of cooperation with foundations, academic centres or other companies. The drug development market benefits from numerous forms of financing such as research grants, sale of shares in a project or company, loans, support from foundations and many others. An important aspect from the project development perspective is also a decision on the exit strategy, i.e. whether and at what stage of project development do the owners aim to commercialise a project and on what financial or ownership terms.
Market ownership structure - Drug discovery and development requires a long-term, passionate and believing view of the investment being undertaken, which is a long and risky process, hence the discussion on whether the current form of market ownership is appropriate to maintain an appropriate level of involvement, resistance to the changing market situation and accept lower margins in the long run. Over the last 20-30 years, a decrease in the productivity of large pharmaceutical companies has been observed, resulting in numerous acquisitions and mergers. At the same time, organisations with different structures, such as foundations, private companies or non-profit organisations, were and are better able to adapt to periods of reduced profits or unstable factors of the market environment.
Creation of biotechnology companies and start-ups - these two forms of scientific organisations are often a solution for people who do not have the right career paths in large pharmaceutical companies or subsidiaries. Smaller organisations tend to be more flexible and more agile in decision-making, allowing for faster progress in the implementation of scientific ideas. Biotechnology companies and start-ups usually focus on a narrow therapeutic area or on selected technologies, which is dictated by a smaller scale of operation, limited budget, but also limited research infrastructure and reliance on external suppliers (outsourcing).
Expiring blockbuster patent protection - many drugs that dominate the market of selected therapeutic areas, so-called blockbusters, lose patent protection, which prompts companies to work on generic drugs that are more likely to be registered and marketed, reducing the number of drug developments.
Early differentiation from competing projects - maintaining competitiveness in long-term and risky projects is a difficult process, one that requires not only focusing on project work, but also tracking and verifying data received in competitive projects. Early implementation of differentiation strategies increases the chances of quickly spotting errors in the design, changing the strategy if possible, and defining the chances of success and maintaining competitiveness in relation to other projects from the market developed for the same molecular purpose
Environmental factors:
Lifestyle of the populations - the concept of the Western way of life appeared in connection with the development of agriculture and industry. A more sedentary mode of functioning, with a less varied diet and in a more polluted environment significantly affects the functioning of the human body. In the context of drug development emphasis is placed on the impact of change on the development of civilisation diseases, such as cardiovascular diseases, obesity, type 2 diabetes, osteoporosis or certain forms of cancer. These diseases are largely associated with a sedentary lifestyle, excessive consumption of food or stimulants, and pose a challenge in the development of drugs, because during the design and development of therapies environmental factors and the interaction of potential therapeutics with substances delivered to the body with food should be taken into account. In addition, the influence of external factors on the human genome, the changes occurring in it and the inheritance of these changes is another dimension and challenge in the development of drugs - an area of science called epigenetics.
Diagnosis and response to an unmet medical need - this area of diagnostics is still developing, allowing for the recognition and isolation of new disease entities. Drug development targeting diseases for which there are no available treatments are more likely to succeed, as there is less competition in these areas, and regulatory requirements are often more lenient. This is to allow the introduction of forms of therapy to the market for patients with unmet medical needs who have no other chance to improve their health situation.
Patient-friendly dose and route of administration - the form of the drug and its dosage should be user-friendly - if possible without the need for additional medical devices to administer them, allowing for matching doses to the patient and intuitive. This is important from the point of view of the production process of the drug, but also important for people who are sick, lonely, disabled or elderly, and for people caring for patients, so as not to cause difficulties in the treatment process. Difficulties in using the drug may result in abandonment of its use or improper use, which affects the appearance of side effects or lack of effectiveness of treatment.
Conclusions
The drug discovery field influences various areas of daily life - starting from the condition of the patients, through the health care system condition, economy of the regions and creation of life style habits of societies. On the other hand, the same factors impact the productivity of drug discovery research. During evaluation, relationships between the factors were recognised, and it was observed that Complexity of disease and pathophysiology is correlated with Molecular target selection, Biomarkers availability and Selection of patient populations, while knowledge about the disease and its progression provides the basis for the project design and approach. This, in many cases, is correlated with the network of Scientific cooperations performed by an organisation. Pathophysiology and new diseases are related to the Lifestyle of the population and often driven by the daily choices of patients.
A relation between the Selection of a molecular target, Selection of a compound for further development and Selection of patient populations can be seen when these three factors influence the overall process of developing new therapies. It is the choices made in these three areas that define the further path of development of a new drug and, consequently, define the probability of successful registration of the drug. Many months, and often years, pass between the decision on the selection of the molecular target and the selection of the lead compound, or later - the selection of the patient population, where significant investments are made. This decision requires the involvement of key stakeholders in the project initiation decision followed by Appropriate preparation for project management, bearing in mind the long drug discovery process. As a result, companies working in the industry have to introduce remediation and risk reduction strategies using a platform approach, introducing Novel methods and design strategies and Early differentiation from competing projects into the ways of working. Companies need also to select projects that they are capable to perform, which is related to Ensuring a long-term and flexible financing strategy as well as Expiring blockbuster patent protection and the Market ownership structure, as well as the way in how companies and research are financed. The market structure is driven by the number of big and small companies defined as Creation of biotechnology companies and start-ups. As discussed above, small companies are mainly created by scientists who are employees not satisfied with the available Career paths for researchers in existing companies.
In terms of scientific cooperation, it was observed that this factor is highly related to Appropriate, complete and clear project documentation, which influences Consultation with clinicians and clinical centres supporting projects with knowledge on the disease mechanisms, patients' conditions or samples from the patients needed in laboratory research.
Multidimensional relations between the recognised factors give insights into the valuation and decision-making process related to the field of drug discovery, and also supports the adjustment of project management and the business approach to address globalisation of the markets, competitiveness and needs of patients waiting for new and safe forms of treatment. The results of the study also suggest that further work on defining key factors and their correlations is required to address the needs of the multidimensional ecosystem of drug discovery research projects.
Acknowledgements
This publication was co-financed/financed from the subsidy granted to the Krakow University of Economics - Project no. 040/SD/2022/PRO.
References
- Antonijevic, Z. (Ed.). (2015). Optimization of pharmaceutical R&D programs and portfolios. Design and investment strategy. Springer International Publishing AG.
- Berdigaliyev, N., & Aljofan, M. (2020). An overview of drug discovery and development. Future Medicinal Chemistry, 12(10). https://doi.org/10.4155/fmc-2019-0307
- Bode-Greuel, K., & Nickisch, K. (2008). Value-driven project and portfolio management in the pharmaceutical industry: Drug discovery versus drug development - Commonalities and differences in portfolio management practice. Journal of Commercial Biotechnology, 14(4), 307-325. https://link.springer.com/article/10.1057/jcb.2008.6#citeas
- Bukowski, H., & Gierczyński, J. (2019). Przełomowe innowacje farmaceutyczne - doświadczenia, rekomendacje, perspektywy. Instytut Innowacyjna Gospodarka dla INFARMA. https://www.infarma.pl/assets/files/raporty/raport_przelomowe_innowacje_farmaceutyczne.pdfzne.pdf (infarma.pl)
- Chen, J. L., Luo, X. Qiu, H., Mackey, V., Sun, L., & Ouyang, X. (2018). Drug discovery and drug marketing with the critical roles of modern administration. American Journal of Translational Research, 10(12), 4302-4312.
- Chung, T. D. Y., Terry, D. B., & Smith, L. H. (2015). In vitro and in vivo assessment of ADME and PK properties during lead selection and lead optimization - guidelines, benchmarks and rules of thumb. In S. Markossian, A. Grossman, K. Brimacombe, M. Arkin, D. Auld, C. Austin, J. Baell, T. D. Y. Chung, N. P. Coussens, J. L. Dahlin, V. Devanarayan, T. L. Foley, M. Glicksman, K. Gorshkov, J.V. Haas, M.D. Hall, S. Hoare, J. Inglese, P. W. Iversen. X. Xu (Eds.), Assay Guidance Manual [Internet]. The Assay Guidance Manual, National Library of Medicine.
- Cook, D., Brown, D., Alexander , R., March, R., Morgan, P., Satterthwaite,,G., & Pangalos, M. N. (2014). Lesson learned from the fate of Astra Zeneca's drug pipeline: a five-dimension framework. Nature Reviews Drug Discovery, 13(6), 419-431. https://doi.org/10.1038/nrd4309
- Czech, M. (2022). Leki. Od odkrycia do pacjenta. Wymiary zarządzania systemem. Polskie Wydawnictwo Ekonomiczne.
- DiMasi, J. A. (2000). New drug innovation and pharmaceutical industry structure: trends in the output of pharmaceutical firms. Drug Inform Journal, 34, 1169-1194. https://doi.org/10.1177/009286150003400425
- EvaluatePharma. (2018). World Preview 2018, Outlook to 2024 (11th ed.). https://www.evaluate.com/sites/default/files/media/download-files/WP2018.pdf
- EvaluatePharma. (2019). World Preview 2019, Outlook to 2024 (12th ed.). https://info.evaluate.com/rs/607-YGS-364/images/EvaluatePharma_World_Preview_2019.pdf
- FDA. (2018). The drug development process. https://www.fda.gov/patients/learn-about-drug-and-device-approvals/drug-development-process
- FDA. (2023). Novel drug approvals for 2022. https://www.fda.gov/drugs/new-drugs-fda-cders-new-molecular-entities-and-new-therapeutic-biological-products/novel-drug-approvals-2022
- Hingorani, A. D., Kuan, V., Finan, C. Kruger, F. A., Gaulton, A., Chopade, S., Sofat, R., MacAllister, R. J., Overington, J. P., Hemingway, H., Denaxas, S., Prieto, D., & Casas, J. P. (2019). Improving the odds of drug development success through human genomics: modelling study. Scientific Reports, 9, 18911. https://doi.org/10.1038/s41598-019-54849-w
- Kiriiri, G. K., Njogu P. M., & Mwangi, A. N. (2020). Exploring different approaches to improve the success of drug discovery and development projects: a review. Future Journal of Pharmaceutical Sciences, 6(27). https://doi.org/10.1186/s43094-020-00047-9
- Liberti, L., Breckenridge, A., Eichler, H. G., Peterson, R., McAuslane, N., & Walker, S. (2010). Expediting patients' access to medicines by improving the predictability of drug development and the regulatory approval process. Clinical Pharmacology & Therapeutics, 87(1), 27-31. https://doi.org/10.1038/clpt.2009.179
- Light, D. W. (2012). Pharmaceutical R&D. The Politics of Medicines (e-Encyclopaedia). https://haiweb.org/encyclopaedia/pharmaceutical-research-and-development/
- Mikulic, M. Total global pharmaceutical R&D spending 2014-2028, Statistica.com, October 2022.
- Mikulic, M. (2022). Worldwide generic prescription drug sales 2012-2026. https://www.statista.com/statistics/309411/global-total-generic-prescription-drug-revenue/
- Mingus, N. (2002). Alpha teach yourself project management in 24 hours. Alpha.
- Mullard, A. (2023, January 3). 2022 FDA approvals. Nature Reviews Drug Discovery, 22, 83-88. https://doi.org/10.1038/d41573-023-00001-3
- Ng, R. (2015). Drugs: From discovery to approval (3rd ed.). John Wiley & Sons Inc.
- Paul, S. M., Mytelka, S., Dunwiddie, Ch. T., Persinger, Ch. C., Munos, B. H., Lindborg, S. R., & Schacht, A. L. (2010). How to improve R&D productivity: the pharmaceutical industry's grand challenge. Nature Reviews Drug Discovery, 9, 203-214 (2010). https://doi.org/10.1038/nrd3078
- Petro, Y., & Gardiner, P. (2015). An investigation of the influence of organizational design on project portfolio success, effectiveness and business efficiency for project-based organizations. International Journal of Project Management, 33(8), 1717-1729. https://doi.org/10.1016/j.ijproman.2015.08.004
- Project Management Institute. (2021). PMBOK guide. A guide to the Project Management Body of Knowledge (7th ed.).
- Poduri, R. (Ed.). (2021). Drug discovery and development, from targets and molecules to medicines. Springer.
- Rydzewski, R. (2008). Real world drug discovery. A chemist's guide to biotech and pharmaceutical research. Elsavier.
- Sagar, G. V. (2017). Pharmaceutical industrial management (2nd ed.). BSP Books Pvt. LTD.
- Schuhmacher, A., Gassmann, O., Hinder, M., & Kuss, M. (2021). The present and future of project management in pharmaceutical R&D. Drug Discovery Today, 26(1). https://doi.org/10.1016/j.drudis.2020.07.020
- Schuhmacher, A., Gassmann, O., Bieniok, D., Hinder, M., & Hartl, D. (2022). Open innovation: A paradigm shift in pharma R&D? Drug Discovery Today, 27(9), 2395-2405. https://doi.org/10.1016/j.drudis.2022.05.018
- Schuhmacher, A., Hinder M., & Gassmann O. (Eds.). (2016). Value creation in the pharmaceutical industry. Willey-VCH. https://doi.org/10.1002/cmdc.201700797
- Stalder, J. (2022). Project management for drug developers. CRC Press Inc.
- Tranfield, D., Denyer, D., & Smart, P. (2003). Towards a methodology for developing evidence-informed management knowledge by means of systematic review. British Journal Management, 14(3), 207-222. https://doi.org/10.1111/1467-8551.00375
- Wallmark, B. (2016). Success factors in drug discovery and development. Advances in Precision Medicine, 1(2), 1-5. http://dx.doi.org/10.18063/APM.2016.02.002
- Yeung, A. W. K., Atanasov, A. G., Sheridan, H., Klager, E., Eibensteiner, F., Völkl-Kernsock, S., Kletecka-Pulker, M., Willschke, H., Schaden, E. (2021). Open innovation in medical and pharmaceutical research: A literature landscape analysis. Frontiers in Pharmacology, 11. https://doi.org/10.3389/fphar.2020.587526
- Zurdo, J. (2013). Developability assessment as an early de-risking tool for biopharmaceutical development. Pharmaceutical Bioprocessing, 1(1), 29-50.



 https://orcid.org/0000-0003-2266-9381
https://orcid.org/0000-0003-2266-9381